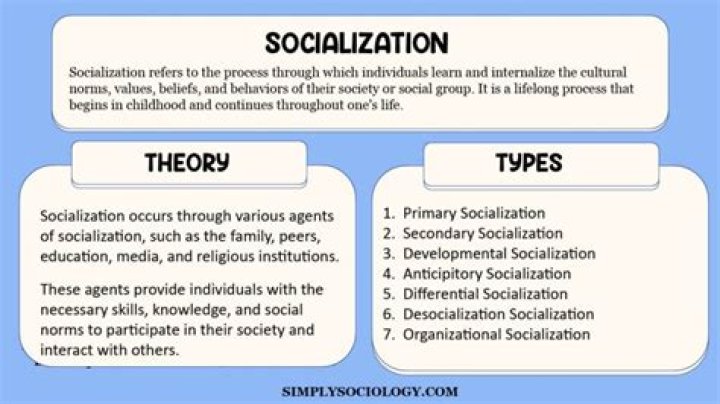
Which conformation of cyclohexane is least stable

William Smith
Updated on April 05, 2026
The least stable conformation of cyclohexane is half chair conformation.
Which of the following conformation of cyclohexane is the least stable?
The least stable conformation of cyclohexane is half chair conformation.
Which conformation of cyclohexane is more unstable?
Answer: Half chair form. Explanation: It is the conformer of cyclohexane having the highest enegry in the energy profile diagram of cyclohexane.
Which conformation is least stable?
…with respect to the other—the eclipsed conformation is the least stable, and the staggered conformation is the most stable. The eclipsed conformation is said to suffer torsional strain because of repulsive forces between electron pairs in the C―H bonds of adjacent carbons.Which cyclohexane conformer is most stable?
The most stable conformation of cyclohexane is the chair form shown to the right. The C-C-C bonds are very close to 109.5o, so it is almost free of angle strain. It is also a fully staggered conformation and so is free of torsional strain.
Which of the following is correct order of stability of cyclohexane?
Among the important confermers of cyclohexane, the order of stability is half-chair < boat<twist boat< chair.
Which of the following cyclohexane conformations has the most energy is the least stable )? A chair B half chair C Boat D twist boat?
Boat conformation is the least stable, with the highest energy, has steric hindrance between the two equatorial hydrogens on carbon 1 and carbon 4, and has torsional tension because each bond almost completely ellipses other bonds in the Newman projection.
Which conformation has the highest and lowest torsional strain?
Staggered form has the least torsional strain and the eclipsed form has the maximum torsional strain.Which of the following is least stable eclipsed conformation?
The fully eclipsed conformation of n-butane is least stable due to the presence of. No strain is present in the molecule.
Which of the following conformation is most stable for methyl cyclohexane?The most stable conformation of methylcyclohexane is the chair conformation in which the methyl group is equatorial. The alternative chair conformation, in which the methyl group is axial, is 7.3 kJ/mol higher in energy.
Article first time published onWhy is the chair conformation of cyclohexane more stable than the boat?
Answer: Chair conformation of cyclohexane is more stable than boat form because in chair conformaion the C-H bonds are equally axial and equatorial, i.e., out of twelve C-H bonds, six are axial and six are equatorial and each carbon has one axial and one equatorial C-H bond.
How do you know which chair conformation is more stable?
To Determine Chair Conformation Stability, Add Up The A-Values For Each Axial Substituent. The Lower The Number, The More Stable It is.
Which cyclohexane conformation is more stable and why?
The chair conformation is more stable because it does not have any steric hindrance or steric repulsion between the hydrogen bonds.
Which is more stable cyclohexane or hexane?
Since cyclohexane molecules are closely packed, the molecular interactions are much stronger than that of hexane. Therefore, the energy required to break the bonds and evaporate it is much higher. Hence, cyclohexane has a higher boiling point.
Which cyclohexane conformations has the most energy and the least stable?
half – chair has the MOST energy and is the least stable.
Which one of the following is the most energetic conformation of cyclohexane?
Most stable and least energetic conformation of cyclohexane is half-chair.
Which of the following represents conformation of cyclohexane?
Conformation of cyclohexane that are free of angle strain: Chair conformation is achiral as it possesses a center of symmetry while boat conformation is achiral as it possesses a plane of symmetry. Twist boat conformation is chiral as there is no symmetry element.
Which of the following is correct order of stability?
Sn2+<Ge2+
Why is half chair conformation of cyclohexane least stable?
The chair conformation is staggered about all bonds and therefore there is no torsional strain. Also it doesn’t have any angle strain as compared to all other conformations, thus most stable. The half chair form is least stable due to maximum strain.
Why is half chair conformation least stable?
The chair conformation is spread and staggered at all the bonds therefore there is no torsional strain. All the C−C bond is very close and is free of any angle strain making it most stable conformation. The half chair form is less stable than the chair used to the presence of some strain.
Which of the following is least stable a anti conformation B gauche conformation C staggered conformation D eclipsed conformation?
Answer: d Explanation: Eclipsed conformation is the least stable due to its high torsional energy. The potential energy of n-butane is maximum for eclipsed conformations.
Why is eclipsed conformation less stable?
The ethane molecule depicted here is in the eclipsed conformation. This is the least stable form of ethane. It is least stable because there is a minimum distance between electron densities of C-H bonds, and therefore a maximum amount of electron-electron repulsion. This interaction is called torsional strain.
Which conformers are more stable?
A staggered conformer is more stable than an eclipsed conformer as the latter involves unfavourable energy interactions between atoms.
Which is more stable cyclohexane or cyclopentane?
Cyclopentane is a more stable molecule with a small amount of ring strain, while cyclohexane is able to adopt the perfect geometry of a cycloalkane in which all angles are the ideal 109.5° and no hydrogens are eclipsed; it has no ring strain at all.
Which conformation of cyclopentane is more stable?
The first conformation is more stable. Even though the methyl groups are trans in both models, in the second structure they are eclipsing one another, therefore increasing the strain within the molecule compared to the first structure where the larger methyl groups are anti to one another.
Which is least stable conformation of ethane?
The eclipsed conformation of ethane is less stable than the staggered conformation by 3 kcal/mol.
Which of the following conformation is most stable in monosubstituted cyclohexane?
Monosubstituted Cyclohexanes In the previous section, it was stated that the chair conformation in which the methyl group is equatorial is more stable because it minimizes steric repulsion, and thus the equilibrium favors the more stable conformer. This is true for all monosubstituted cyclohexanes.
Why is the boat conformation less stable?
The boat conformation suffers from torsional strain, making it less stable (higher in energy) than the chair. Steric strain in the boat arises mainly from the repulsion (steric crowding) between the two hydrogens on the ends of the “boat.
Which of the following conformation has highest stability Mcq?
D. The chair form is the most stable conformation of cyclohexane. The C−C−C bonds are very similar to 109.5o , so they are almost free from angular strain.
Which is the more stable of the two chair conformers shown?
Out of two conformations, the one with lower energy is more stable. So, despite having two axial groups, the first conformer is more as two chlorines do not bring as much steric interaction as the methyl group.
Which is more stable cyclohexane or cyclohexene?
Another difference between cyclohexane and cyclohexene is that the cyclohexane is relatively stable, thus, is less reactive while the cyclohexene is relatively unstable, thus, can undergo reactions due to the presence of a double bond in the ring structure.