What does but 1 ENE mean

Gabriel Cooper
Updated on April 09, 2026
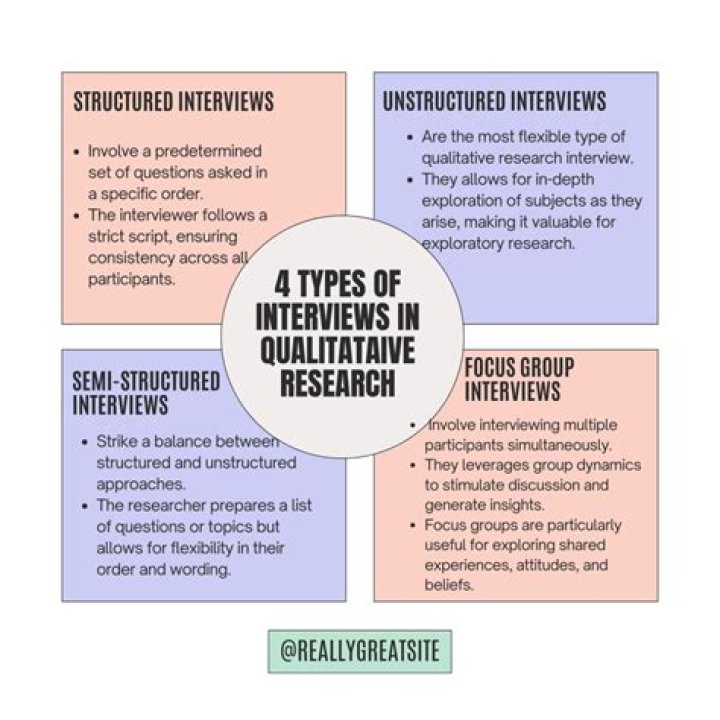
Notice that butene has two different forms called isomers . But-1-ene and but-2-ene have the same molecular formula, but the position of their C=C bond is different. The number in their names show where that bond is located in the molecule.
Why is it called but-1-Ene?
Notice that butene has two different forms called isomers . But-1-ene and but-2-ene have the same molecular formula, but the position of their C=C bond is different. The number in their names show where that bond is located in the molecule.
Is butene the same as but-1-Ene?
Rationale behind it is to have the ability to distinguish between say, But-1-ene & But-2-ene. Otherwise no, there’s no difference.
What's the difference between but-1-ene and But-2-ene?
But-1-ene and But-2 -ene are structural isomers. They differ in the position of double bonds. In but-1-ene the double bond is present in between the 1st carbon and the second. But in But-2-ene, the double bond is present between the 2nd and the 3rd carbon atom.What are the uses of 1-butene?
/1-Butene/ is used for the production of a wide variety of chemicals in the gasoline and rubber processing areas. More than half of the butenes produced world-wide are utilized as alkylate and polymer gasoline. One third is used without any conversion as fuel, mainly as fuel gas or blendstock for gasoline.
What is the same for butene and hexene?
Butene and hexene (strictly speaking: butene-1 and hexene-1) are examples of comonomers that are used during the production of different polyethylene (PE) grades, including roto grades. They do essentially the same thing for PE, but their presence can cause some differences in grade performance.
Is 1-butene toxic?
NamesSignal wordDangerHazard statementsH220 , H221 , H280Precautionary statementsP210 , P377 , P381 , P403 , P410+P403NFPA 704 (fire diamond)1 4 0
How would you distinguish between but-1-ene and but-2-ene by ozonolysis?
The chemical distinction between but-1 ene and but -2 ene can be done by looking at the ozonolysis products. On ozonolysis of but- 1 ene,, will give propanal and formaldehyde. Where as on the ozonolysis of But-2-ene, the product obtained is two moles of ethanal.Is but-1-ene saturated?
But-1-ene is a butene with unsaturation at position 1.
Is but-2-ene saturated?2-butene appears as a colorless liquefied petroleum gas. Asphyxiate gas. Flammability limits in air 1.8-9.7% by volume.
Article first time published onCan you have Prop 1 Ene?
this is the structural formula for prop-1-ene. as name indicates prop used for 3 carbon organic compound and ‘ene’ is used for alkenes so it is a three carbon alkene havine double bond between 1st and 1nd carbon having formula C3H6. this is the structural formula for prop-1-ene.
What type of polymerisation does but-1-ENE undergo?
In a rather low percentage, but-1-ene is used as one of the co-monomers, along with other α-olefins, in the production of high-density polyethylene (HDPE) and linear low-density polyethylene (LLDPE). Butyl rubber is made by cationic polymerisation of isobutylene with between 2 and 7% isoprene.
Are but-1-ene and but 2 ene isomers?
But-1-ene and but-2-ene are called structural isomers because they have: same molecular formula (C4H8) different structural formula (position of double bond is differrent)
How many bonds are there in 1-butene?
Chemical Structure Description The 1-BUTENE molecule contains a total of 11 bond(s) There are 3 non-H bond(s), 1 multiple bond(s), 1 rotatable bond(s) and 1 double bond(s). The 2D chemical structure image of 1-BUTENE is also called skeletal formula, which is the standard notation for organic molecules.
Do geometric isomers exist for but-1-Ene?
but-1-ene. ⚠ Does not have geometrical isomers, even though it has a C=C bond! One of the C atoms in this bond has two identical groups (H atoms) attached.
Is butene 1 liquid or gas?
1-Butene is a flammable, colorless gas which is shipped as a liquefied gas at its vapor pressure of 23.5 psig at 70F.
Is 1-butene polar or nonpolar?
Information on this page: Normal alkane RI, non-polar column, temperature ramp. References.
How do you make isobutylene?
Production. Polymer and chemical grade isobutylene is typically obtained by dehydrating tertiary butyl alcohol (TBA) or catalytic dehydrogenation of isobutane (Catofin or similar processes).
What polymer is made from butene?
Butene can be used as the monomer for polybutene, but this polymer is more expensive than alternatives with shorter carbon chains such as polypropylene. Polybutene is therefore used in more specialized applications. Butenes are more commonly used to make copolymer (mixed with another monomer such as ethylene).
What do Bromines do to alkenes?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot. … This has the effect of ‘saturating’ the molecule, and will turn an alkene into an alkane.
Do alkenes burn with a non smoky flame?
Complete combustion of alkenes produces carbon dioxide and water, provided there is a plentiful supply of oxygen. Incomplete combustion of alkenes occurs where oxygen is limited and produces water, carbon monoxide and carbon (soot). This causes a smoky flame.
What are the first 5 alkenes?
- Ethene (C2H4)
- Propene (C3H6)
- Butene (C4H8)
- Pentene (C5H10)
- Hexene (C6H12)
- Heptene (C7H14)
- Octene (C8H16)
- Nonene (C9H18)
Which of the following reagents can be used to distinguish but 1 ene and but 2 ene?
A. NaNH2 is used to distinguish between 1-butyne and 2-butyne.
How do you distinguish between but 1 YNE and but 2 YNE?
But-1-yne contains acidic hydrogen and hence it reacts with ammonical Cu2Cl2 to form a red precipitate of copper−1−butynide whereas but-2-yne does not react with ammonical Cu2Cl2 as it do not have acidic hydrogen.
How can you tell the difference between Bute 1 and Bute 2 in?
– Basic difference in two compounds is position of double bond. These aldehydes are unreactive to iodoform test and dont give any colour. Acetaldehyde on iodoform test gives yellow colour.
What happens when but-2-ene undergoes catalytic hydrogenation?
But-2-ene (2-butene), CH3CH=CHCH3, is an alkene, an unsaturated hydrocarbon. It will react with hydrogen gas, H2(g), in the presence of a palladium catalyst (Pd). The product of this hydrogenation reaction is also butane, CH3CH2CH2CH3, which is an alkane, a saturated hydrocarbon.
Why does but-2-ene show geometrical isomerism?
Since isomers have the same molecular formula the difference in their properties might be due to different modes of combination of arrangement of atoms within the molecule. … Since the above compound (but-2-ene) has double bonds and is an alkene therefore is a geometrical isomer.
What is but-2-ene used for?
Its main uses are in the production of gasoline (petrol) and butadiene, although some 2-butene is also used to produce the solvent butanone via hydration to 2-butanol followed by oxidation.
Is it propene or Prop 1 Ene?
IUPAC Nameprop-1-eneAlternative NamesPropene 1-Propene 1-Propylene prop-1-eneMolecular FormulaC3H6Molar Mass42.081 g/molInChIInChI=1S/C3H6/c1-3-2/h3H,1H2,2H3
Why is Prop 2 ene not possible?
Prop-2-ene is not possible. Since the numbering of the Carbons goes from the end closest to the double bond the name Prop-2-ene is incorrect.
Is E but 2 ene symmetrical?
The product produced contains a chlorine atom. And the carbon-carbon double bond of the alkene is now a carbon-carbon single bond. … But if we flip the bottom product over, we can see that the two product molecules are in fact the same. This has occurred because but-2-ene is a symmetrical molecule.